
Full article credit: Subnet Summer
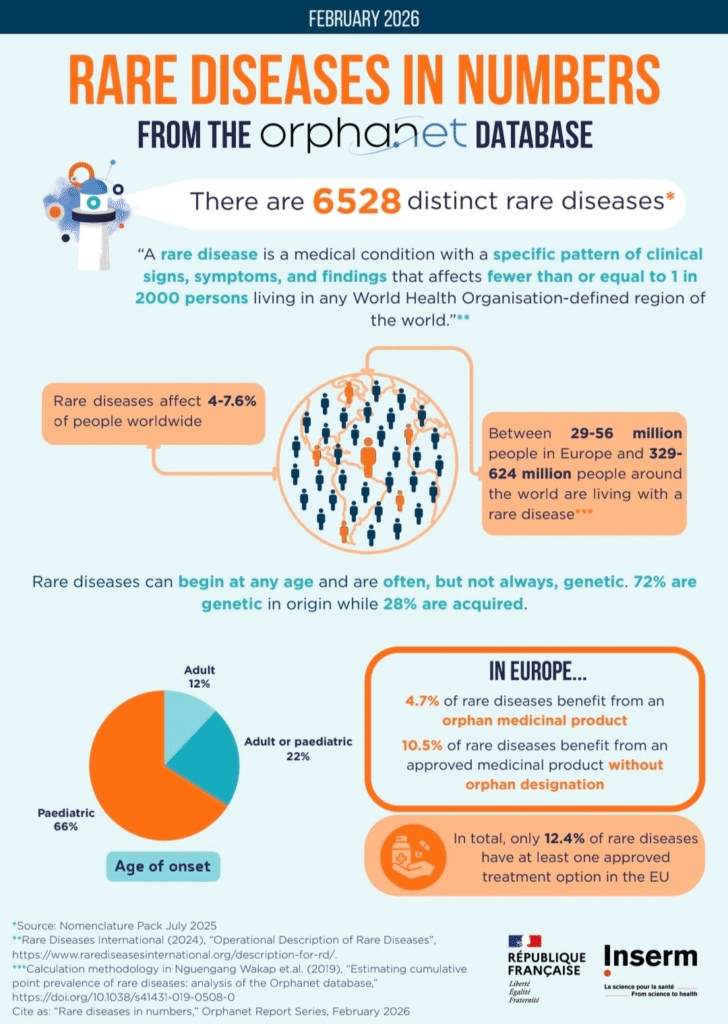
Rare diseases remain one of healthcare’s most persistent blind spots, not because the science is missing, but because the data is fragmented.
For millions of patients worldwide, diagnosis can take years. The challenge is rarely a lack of expertise, but the inability to connect the right information across disconnected systems.
Picture a parent watching their child struggle with unexplained symptoms for years. Test after test, specialist after specialist, only to hear the same frustrating answer: “It’s rare. We don’t have enough data.”
For the more than 300 million people worldwide living with rare diseases, this is not an exception, it is a pattern. And at the center of it is a structural limitation: biomedical data is scattered across hospitals, research institutions, and private organizations, each operating under different constraints, incentives, and access rules.
The Structural Problem Behind Rare Disease Delays:
The challenge is not a shortage of medical knowledge, but a fragmentation of intelligence across the system.
Three constraints consistently emerge:
▫️Data silos persist across healthcare systems:
Critical clinical and genomic data remain locked within institutional boundaries.
▫️AI models are trained on incomplete distributions:
Most systems fail to capture rare or underrepresented biological patterns.
▫️Incentives favor scale, not specificity:
Conditions affecting smaller populations are often deprioritized in traditional R&D pipelines.
The result is predictable, slower discovery cycles, limited reproducibility, and long diagnostic delays for rare conditions.
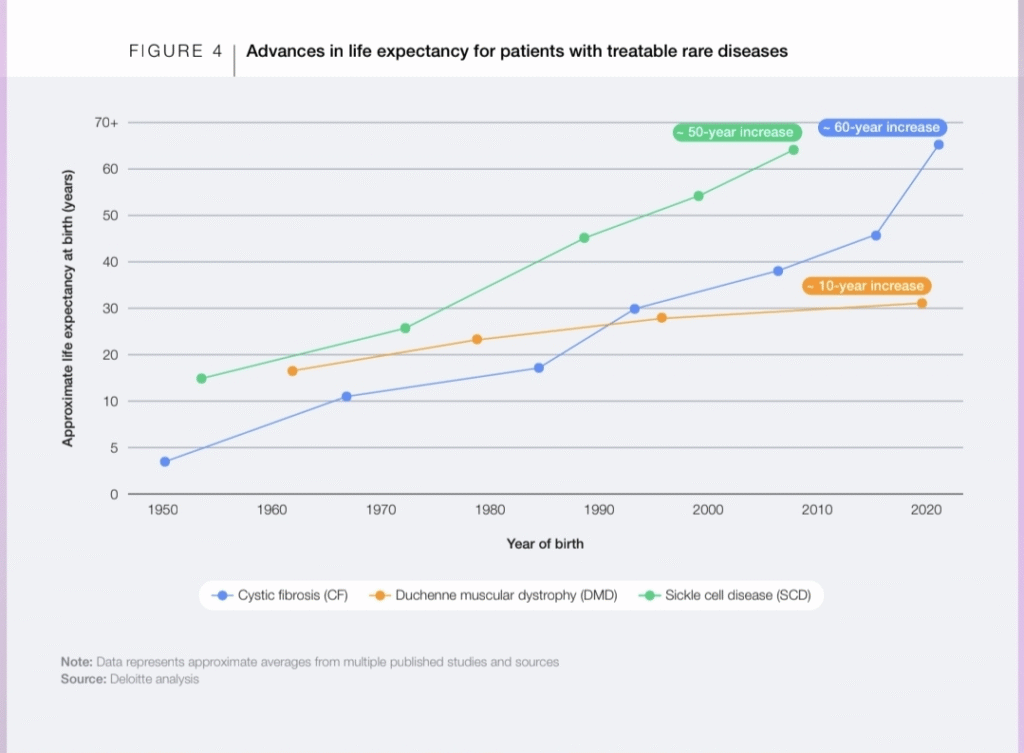
How Bittensor’s Model Offers a Structural Alternative:
Bittensor introduces a different structure for AI development, one built around decentralized coordination rather than centralized control.
Instead of relying on a single organization to train and validate models, Bittensor enables:
▫️independent contributors to build models and compute systems
▫️validators to evaluate outputs based on measurable performance
▫️transparent, incentive-driven reward mechanisms tied to usefulness
This creates a continuous, global competition where intelligence is not stored in one place, but produced across a network.
For fields like rare disease research, this model changes the constraints entirely.
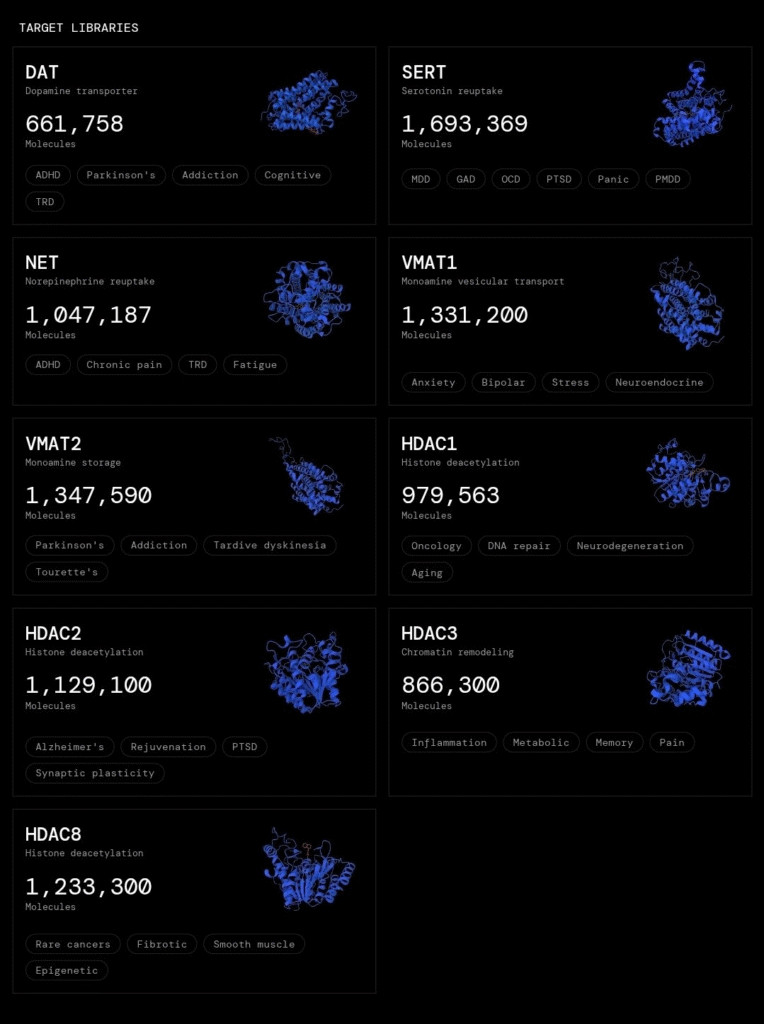
NOVA (SN68): A Practical Layer for Decentralized Drug Discovery:
NOVA, developed by Metanova Labs, is one of the subnets applying this framework directly to drug discovery.
It operates as a decentralized molecular screening system where AI models compete to identify promising compounds against biological targets relevant to real-world diseases.
What makes NOVA significant is not just its design, but its execution:
▫️High-throughput molecular screening at scale:
Thousands of models evaluate vast chemical spaces simultaneously.
▫️Deterministic, verifiable scoring:
Outputs are evaluated through transparent, reproducible mechanisms.
▫️Open participation model:
Researchers and independent contributors globally can contribute and compete.
▫️Alignment with real drug discovery workflows:
Outputs are structured to integrate with downstream medicinal chemistry processes.
Instead of waiting for centralized pipelines to filter hypotheses, NOVA continuously surfaces candidate molecules through decentralized competition.
Why This Matters in Practice:
If systems like NOVA continue to mature and integrate across the broader ecosystem, the implications become tangible:
▫️Faster identification of biological patterns across diverse datasets
▫️Reduced reliance on commercial prioritization in early discovery
▫️Continuous improvement through open, global participation
▫️Increased transparency in AI-driven biomedical research
▫️Expanded access for smaller labs and independent researchers
Importantly, this does not replace hospitals, clinicians, or traditional drug development pipelines. It strengthens the earliest stage of discovery, the stage where most bottlenecks currently exist.
Looking Ahead:
Subnets like NOVA demonstrate that Bittensor can deliver production-grade scientific intelligence today. As the broader network grows, with more specialized compute, secure data contribution mechanisms, and integration between subnets, we move closer to a living, decentralized knowledge layer for rare diseases. One that improves daily because thousands of independent contributors are aligned around a common goal: better answers, delivered faster.
It is still early, but the direction is promising. For the millions of patients and families still waiting, that progress matters.
Final Thoughts:
The next meaningful breakthrough for a rare condition may not come from a single well-funded lab. It could emerge from a global network of minds and machines working openly, rewarded for getting closer to the truth.
That is the quiet but powerful direction Bittensor subnets are exploring in DeSci. We’re excited to follow and support how it develops.
Enjoyed this article? Join our newsletter
Get the latest TAO & Bittensor news straight to your inbox.
We respect your privacy. Unsubscribe anytime.




Be the first to comment